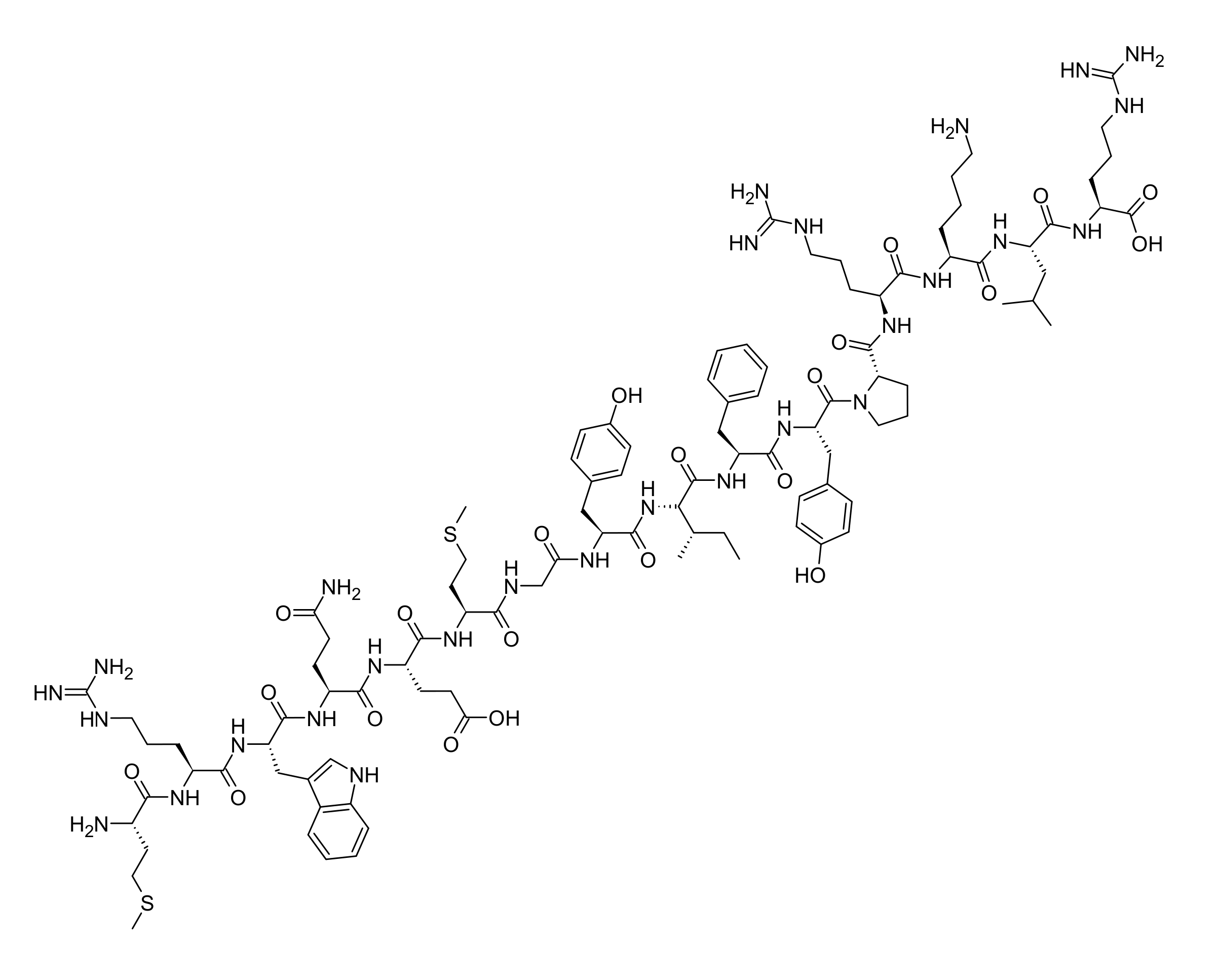
Studies report that MOTS-c interferes with the folate / methionine cycle, raising intracellular AICAR and allosterically activating AMP-activated protein kinase (AMPK) — primarily in skeletal muscle. Secondarily, under metabolic or oxidative stress, MOTS-c translocates from the cytoplasm into the nucleus, where it modulates stress-responsive transcription. MOTS-c is positioned as a retrograde mitochondria-to-nucleus bioregulator coordinating cellular bioenergetics with whole-body glucose handling. Observed in research settings; for research use only.
MOTS-c was the first member of the MDP class to be assigned a defined systemic metabolic role. Its discovery established that mtDNA encodes bioactive peptides via small ORFs in the rRNA genes — MOTS-c on MT-RNR1 (12S rRNA), and humanin and SHLP1-6 on MT-RNR2 (16S rRNA) (Kim/Yen 2021, GeroScience, PMID 32910336). In the original discovery paper (Lee 2015, Cell Metab, PMID 25738459), intraperitoneal MOTS-c administration in mice prevented age-dependent and high-fat-diet-induced insulin resistance and diet-induced obesity, with improved glucose tolerance and reduced fat mass after high-fat feeding — mechanistically mapped to skeletal-muscle AMPK activation via folate-cycle inhibition. Studies report that under metabolic or oxidative stress, MOTS-c translocates from the cytoplasm into the nucleus, where it binds chromatin-associated factors and shifts transcriptional programs toward antioxidant defense and metabolic adaptation (Kim 2018, PMID 29983246). In skeletal muscle, MOTS-c acts as an exercise mimetic — endogenous expression rises after physical activity and exogenous administration improves metabolic flexibility and physical performance in mouse models (Reynolds 2021, PMID 33473109). Important editorial scoping: claims about human pharmacology rest predominantly on observational studies of endogenous MOTS-c (post-exercise plasma levels, allele variants); peer-reviewed human exposure data on **synthetic** MOTS-c administration as a research peptide does not exist, and the only in vivo human Phase 1 dataset is for **CB4211**, a stabilized analog (NCT03998514) — not the natural 16-aa peptide. Mechanism statements here are therefore intentionally hedged — studies report, observed in research settings — and never framed as established human pharmacology. For research use only.