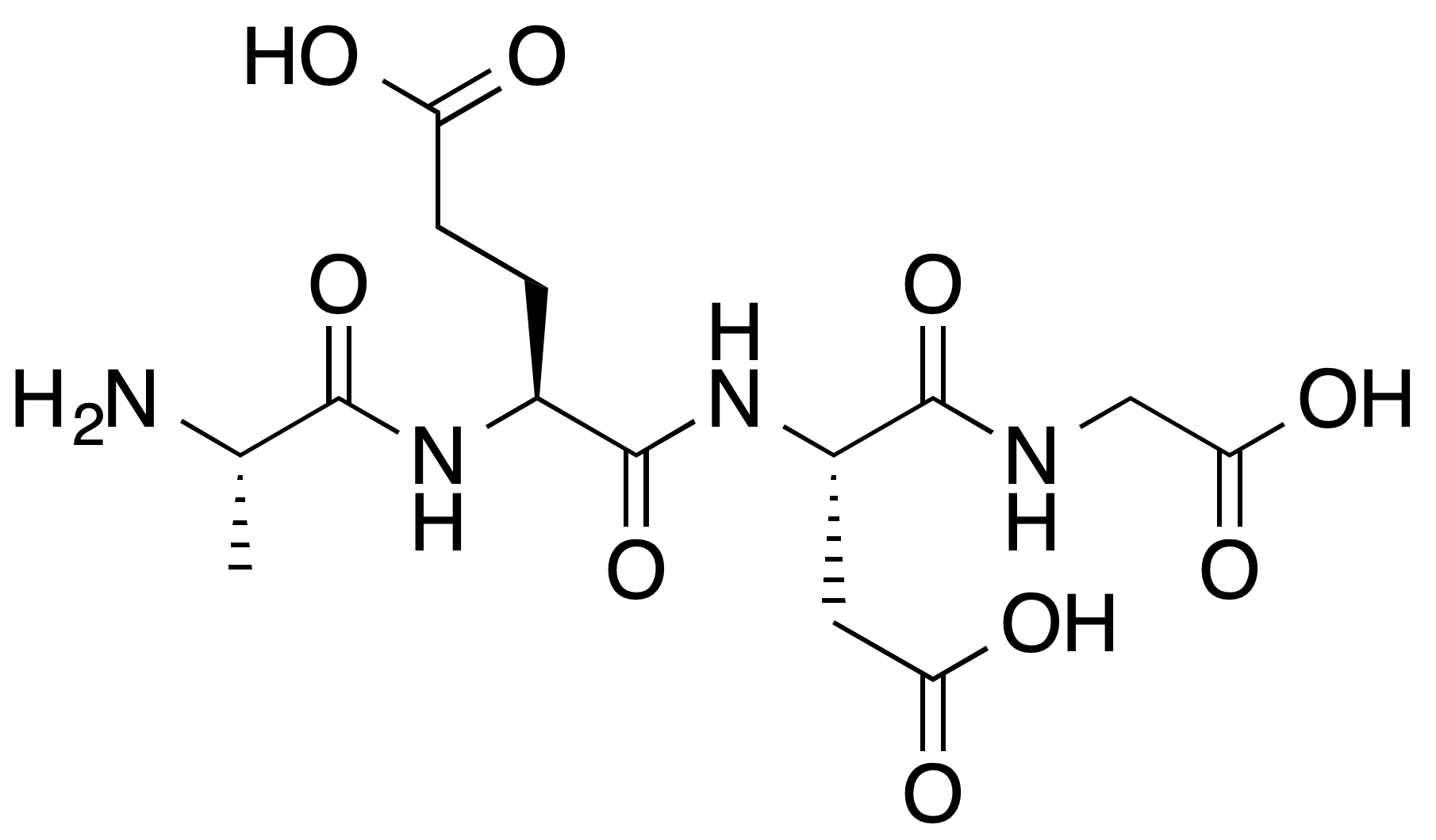
Epitalon (AEDG) is hypothesised to act as a peptide bioregulator that activates telomerase (TERT) in somatic cells in vitro and to modulate pineal melatonin output, with secondary epigenetic effects proposed via direct binding to linker histones H1/3 and H1/6 and selective gene-expression activation. Systemic telomerase activation in living humans has not been demonstrated in peer-reviewed literature.
The mechanistic literature on Epitalon is largely inferred from in vitro fibroblast experiments and rodent studies and is NOT validated by clinical-grade pharmacokinetic or target-engagement data in humans. Studies report that the seminal Khavinson 2003 paper (Bull Exp Biol Med, PMID 12937682) added Epithalon directly to telomerase-negative human fetal fibroblast cultures and observed re-expression of the catalytic TERT subunit, telomerase activity, and continued cell division beyond the Hayflick limit (passage 44+ vs. control halt at passage 34) — an in vitro finding, NOT evidence of human in vivo telomerase reactivation. The 2025 independent in vitro study from Brunel University London / Royal Brompton confirmed dose-dependent telomere extension in normal fibroblasts and epithelial cells via hTERT upregulation, while observing ALT-pathway lengthening in breast-cancer cell lines — the first clearly non-Khavinson-affiliated replication of the core telomere finding, with a concurrent safety flag (Al-Dulaimi 2025, PMID 40908429). The 2025 Polish review by Araj et al. (Int J Mol Sci, PMID 40141333) explicitly concludes that "it remains uncertain whether these are the sole mechanisms of action" and that "information regarding critical issues about this peptide's safety is missing". No peer-reviewed human pharmacodynamic study has demonstrated systemic telomerase activation in vivo from peripheral Epitalon administration. Mechanism statements here are therefore intentionally hedged — studies report, proposed pathways, observed in vitro — and never framed as established human pharmacology. Anti-aging marketing that promotes Epitalon as a human telomerase activator overstates what the published evidence actually shows.