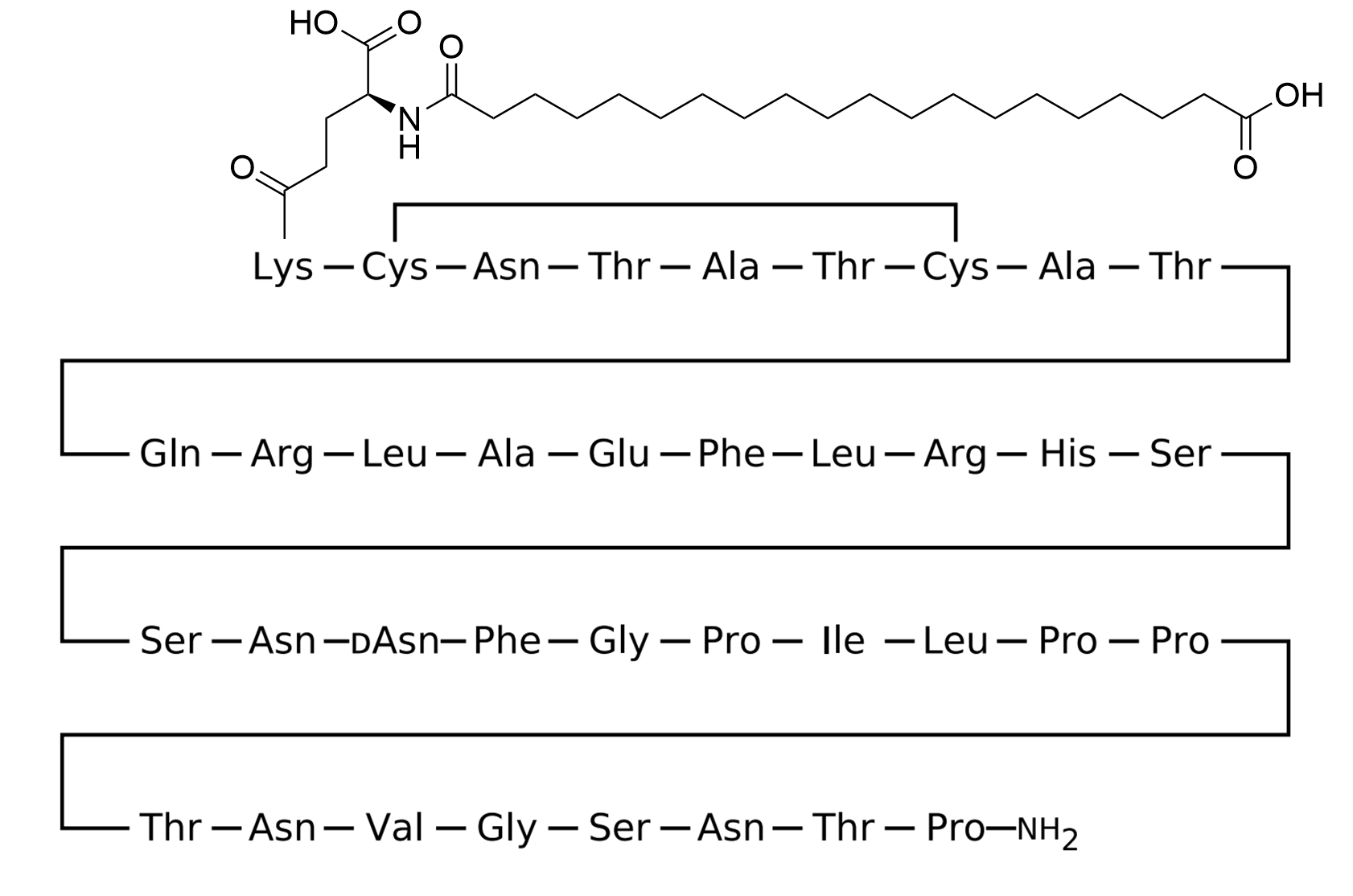
CagriSema couples dual amylin/calcitonin receptor agonism (via cagrilintide) with GLP-1 receptor agonism (via semaglutide), engaging two complementary central and peripheral satiety pathways at once.
The pharmacological rationale for combining the two classes rests on the observation that GLP-1 and amylin receptor agonism act on partially distinct hindbrain and hypothalamic circuits that nevertheless converge on appetite suppression and reduced energy intake. The Phase 2 trial in T2D (Frías et al., Lancet 2023) reported a 32-week body-weight change of −15.6% with CagriSema versus −5.1% with semaglutide alone and −8.1% with cagrilintide alone. In the Phase 3 REDEFINE 1 trial (Garvey et al., NEJM 2025), the 68-week mean body-weight reduction was −20.4% with CagriSema versus −14.9% with semaglutide 2.4 mg, −11.5% with cagrilintide 2.4 mg, and −3.0% with placebo (treatment-policy estimand). These data quantify the additive benefit of dual-pathway engagement in obesity without diabetes. Structural-pharmacology elucidation of cagrilintide binding to calcitonin and amylin receptors was published by Cao et al. (Nat Commun 2025). The ~7-day half-life of each component is achieved by non-covalent serum-albumin binding through the C20 di-acid acylation (cagrilintide) and the C18 di-acid acylation (semaglutide), respectively.