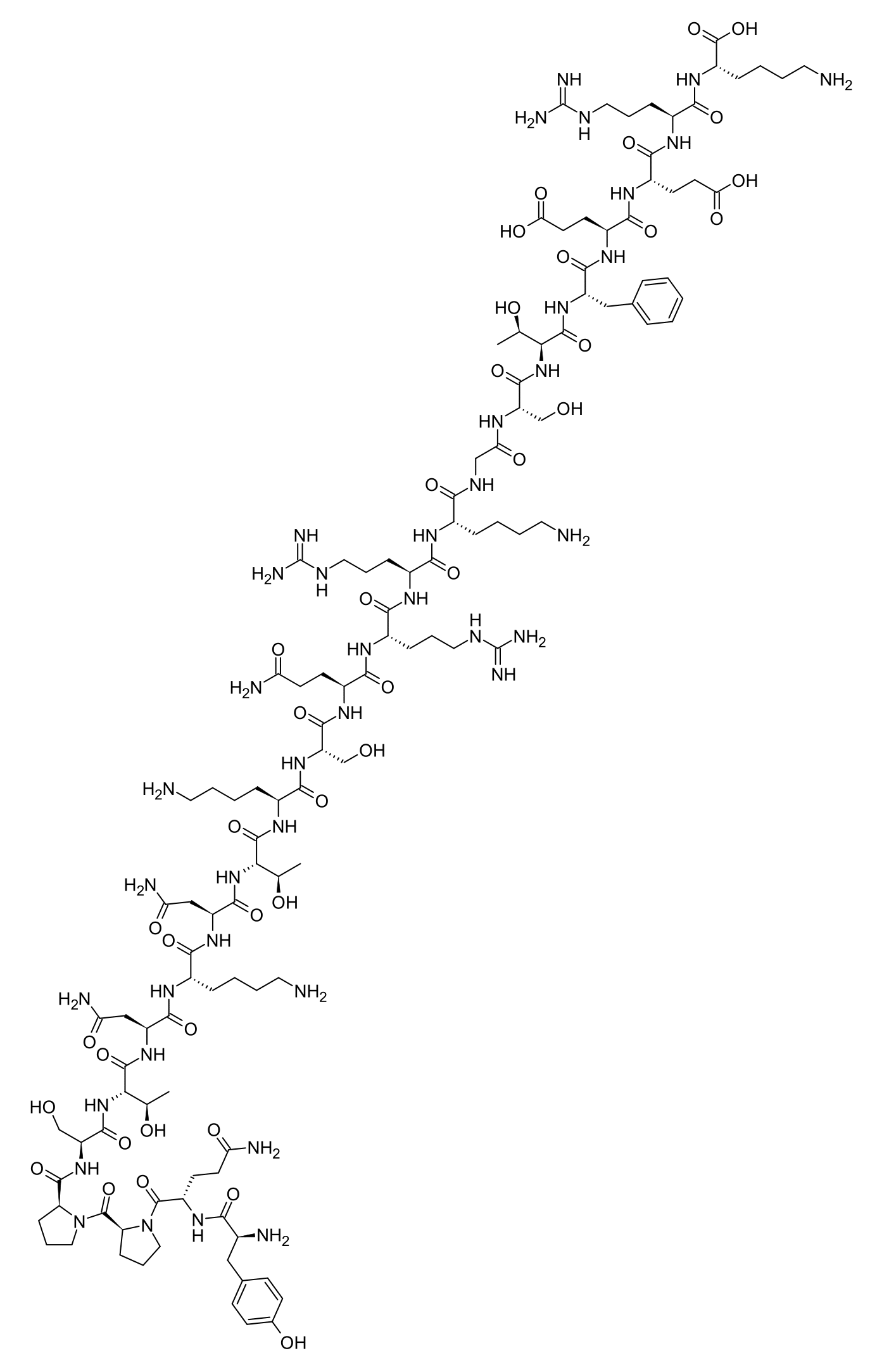
Studies report that the MGF E-peptide (the active principle of PEG-MGF) is a 24-amino-acid peptide rapidly upregulated in mechanically loaded skeletal muscle, reported in preclinical work to promote myoblast proliferation, satellite-cell activation and muscle regeneration. The receptor for the MGF E-peptide is NOT definitively identified as of 2026; several studies report IGF-1R-INDEPENDENT activity (Mills 2007 PMID 17442731), while other studies report IGF-1R-dependent effects (Brisson & Barton 2012 PMID 22802461) — the mechanism is an OPEN question. PEGylation is intended to extend the very short native plasma half-life (~5–7 min; Hill & Goldspink 2003 PMID 12605581); no peer-reviewed human PK characterisation of PEG-MGF exists. Observed in research settings; for research use only.
The biochemistry of PEG-MGF is dominated by two key points. FIRST — the identity of the active substance: the active principle is the 24-amino-acid MGF E-peptide (sequence YQPPSTNKNTKSQRRKGSTFEERK), the C-terminal E-domain peptide of the IGF-1Ec splice variant of human IGF-1 (UniProt P05019 isoform 4 / Ec). Mechanical loading of skeletal muscle (stretch, resistance training) rapidly increases expression of the IGF-1Ec splice isoform at the expense of the systemic IGF-1Ea isoform (Goldspink 1999 PMC1467788; Yang & Goldspink 2002 PMID 12270704); after post-translational processing of the IGF-1Ec pre-pro-hormone, the mature 70-aa IGF-1 chain and the 24-aa MGF E-peptide separate and act as DIFFERENT molecules. This is the page's principal editorial point: MGF is NOT IGF-1, MGF is NOT a "variant", "truncation" or "extension" of IGF-1, but a DIFFERENT peptide produced from the same gene via alternative splicing and processing. SECOND — the open mechanism question: studies report IGF-1R-INDEPENDENT activity for the MGF E-peptide (Mills 2007 PMID 17442731 — the synthetic E-peptide does NOT bind the IGF-1 receptor, and several Goldspink-lineage mechanism claims could NOT be reproduced), while other studies report IGF-1R-dependent effects (Brisson & Barton 2012 PMID 22802461). The definitive receptor for the MGF E-peptide therefore remains UNIDENTIFIED as of 2026. This is an OPEN mechanistic question; the page uses phrasing such as "studies report" and "the receptor for the MGF E-peptide is not definitively identified" and never frames direct IGF-1-receptor agonism as established fact. PEGylation addresses the very short native plasma half-life of ~5–7 minutes (Hill & Goldspink 2003 PMID 12605581); vendors typically prefer 5 kDa or 20 kDa (branched) PEG conjugated at the N-terminal Tyr¹ residue. Vendors claim that PEG-MGF circulates "several days" in humans; however, NO peer-reviewed human PK study of PEG-MGF has been published — the exact human half-life is not characterised. Anti-PEG antibodies are a documented immunogenicity phenomenon for PEGylated drugs (e.g., pegloticase, peginesatide) that has not been studied for PEG-MGF specifically — a theoretical safety concern for chronic / repeated dosing. Observed in research settings; for research use only.